我們為以下罕見疾病提供基因檢測服務:
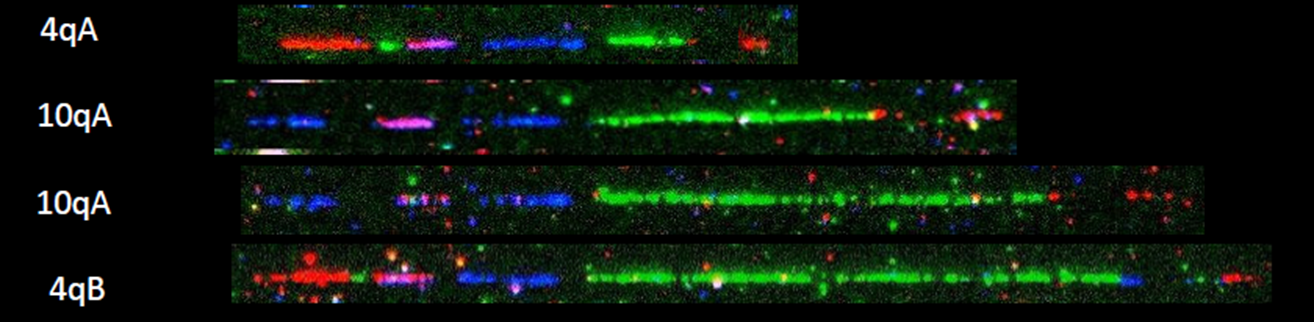
面肩胛肱型肌營養不良症 (FSHD)
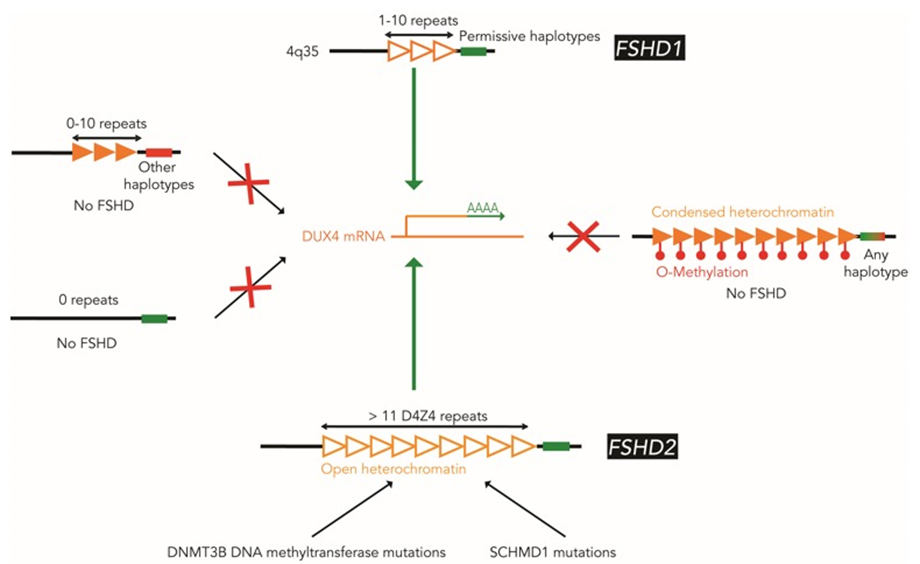
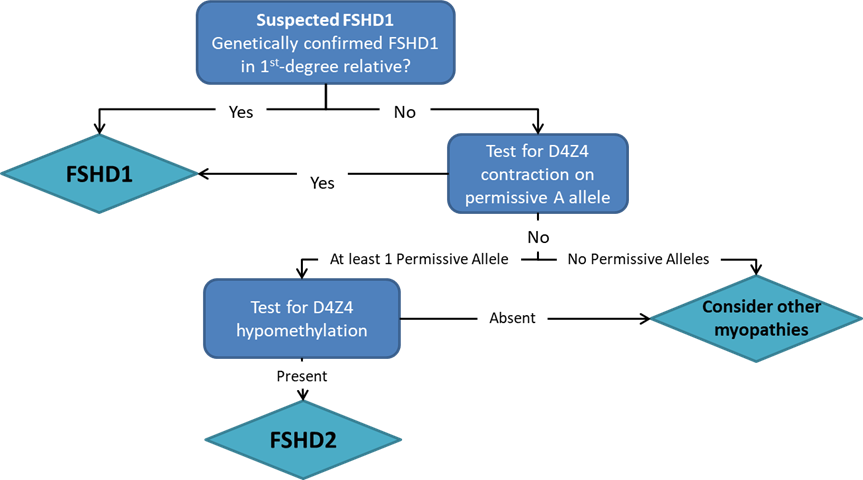
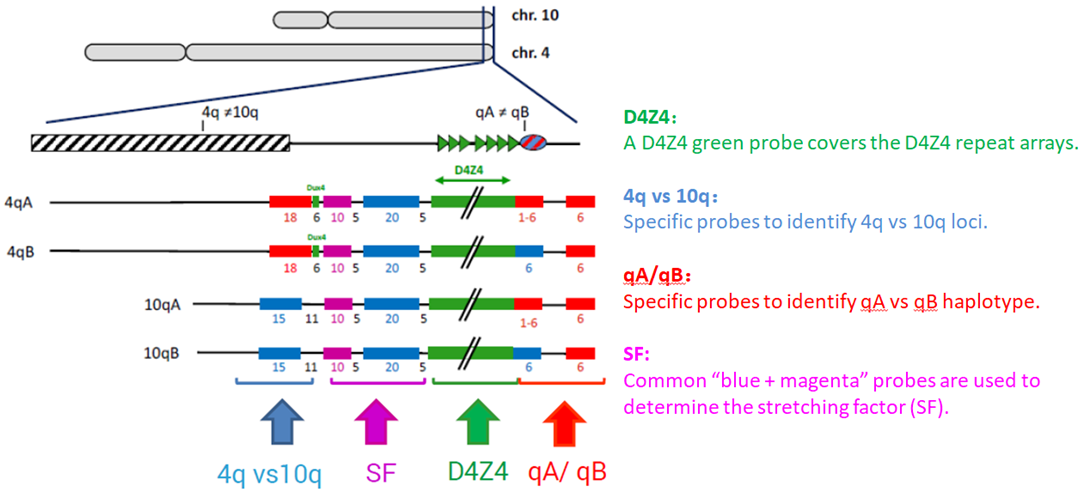
Pangenia Genomics provides the genetic testing of FHSD1 based on the Molecular Combing and Genomic Morse Code technology. This method uses specific fluorescent probes to hybridize with genomic DNA molecules (extracted from fresh blood samples) linearly stretched on coverslips. The fluorescent signals can then be visualized to distinguish the length (number of D4Z4 repeat units) and the type (haplotype A or B) of the D4Z4 loci. Most affected individuals have fewer than 10 repeats and the 4qA allele.
了解更多
杜興氏肌肉萎縮症 (DMD) 和貝克型肌肉萎縮症 (BMD)
Pangenia Genomics offers deletion/duplication analysis to accurately detect the number of copies of the DMD gene, i.e. find out if the gene or part of the gene is deleted or duplicated. It can be used for carrier screening as well.
了解更多
脊髓性肌肉萎縮症(SMA)
Pangenia Genomics offers deletion/duplication analysis in order to accurately detect deletions and duplications in the SMN1 gene. It can be used for carrier screening as well.
了解更多
腓骨肌萎縮症 (CMT)
Pangenia Genomics offers an in vitro diagnostic (IVD) test for the detection of deletions or duplications in the PMP22, MPZ and GJB1 genes for the diagnosis of Charcot-Marie-Tooth disease type 1 (CMT1) disease. The test can be used to confirm the cause and diagnosis for hereditary neuropathy with liability to pressure palsies (HNPP) as well.
了解更多